Main elements of antibody-drug conjugate (ADC): Antibodies and their targets
Virus-like particles (VLP) Platforms for immunogens, vaccines and drug carriers
Antibody-drug Conjugate (ADC): Pre-made ADC benchmark, MOA, Production and QC
Neutralizing antibodies of virus (SARS2, HIV, HBV, Rabies, RSV, Ebola, Influenza)
Immunoglobulin Fc receptors for Fc&Fc Receptor binding assay
ILIBRA-HuEasy Monoclonal antibody (mab) humanization service (fully humanized ab)
Single domain antibody (Nanobody)
SOCAIL MEDIA

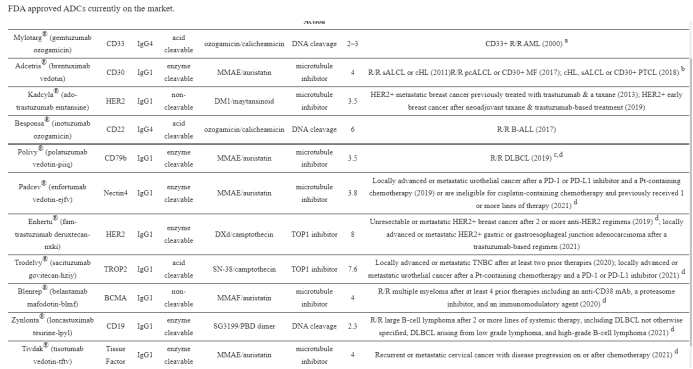
1. Mylotarg® (gemtuzumab ozogamicin) from Wyeth/Pfizer was the first ADC to reach the market. It is composed of a recombinant humanized anti-CD33 mAb (IgG4κ antibody hP67.6) covalently attached to a calicheamicin derived payload (N-acetyl-γ-calicheamicin 1,2-dimethyl hydrazine dichloride) via a pH-sensitive hydrazone linker.
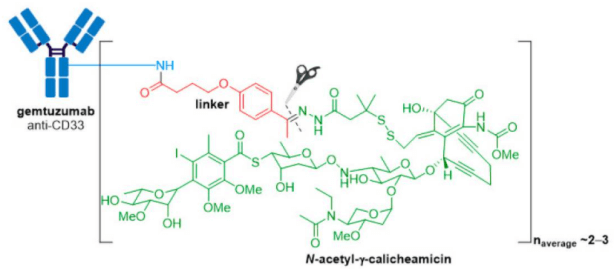
2. Adcetris® (brentuximab vedotin) from Seagen (formerly Seattle Genetics), containing a CD30-specific mAb conjugated to monomethyl auristatin E (MMAE), received FDA approval in 2011, making it the second ADC to enter the oncology market.
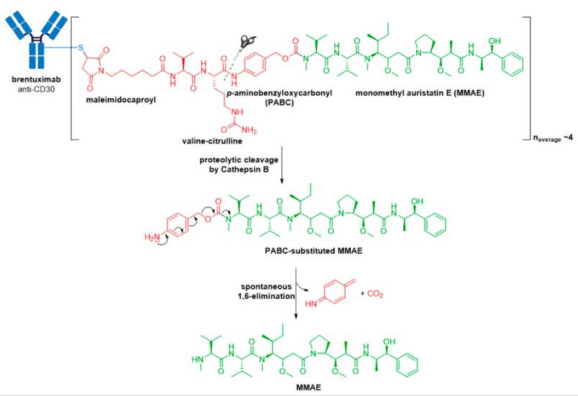
3. In 2013, Kadcyla® (ado-trastuzumab emtansine), developed and marketed by Genentech/Roche, revolutionized the field of ADCs by becoming the first ADC approved for the treatment of solid tumors. It is indicated as an adjuvant (after surgery) treatment for HER2+ early breast cancer in patients who previously received trastuzumab (Herceptin®) and a taxane, separately or in combination
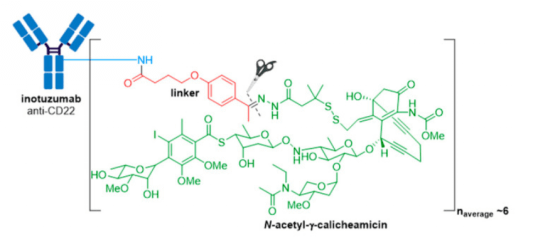
4. Besponsa® (inotuzmab ozogamicin (Pfizer/Wyeth)) obtained FDA approval in 2017 and is directed against CD22+ B-cell acute lymphoblastic leukemia (B-ALL). The first difference lies in the mAb and thus the antigen target and cancer indication. The recombinant humanized monoclonal IgG4 antibody (G544) employed in Besponsa® is selective for CD22 expressed on B cells in all patients with mature B-ALL, and >90% of patients with precursor B-ALL. n.
5. Polivy and Padcev Polivy® is an anti-CD79b ADC developed by Genentech/Roche using a proprietary technology developed by Seagen. It is indicated in combination with bendamustine and rituximab for treatment of adults with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), an aggressive type of non-Hodgkin lymphoma, who have received at least two prior therapies. This indication was granted accelerated approval based on a complete response rate. Polivy® has an approximate DAR of 3.5 molecules of MMAE attached to each antibody.
Padcev®, produced and marketed by Astellas Pharma Inc. and Seagen is a Nectin4-directed ADC. It was first granted accelerated approval in 2019 for treatment of adults with locally advanced or metastatic urothelial cancer who have previously received a programmed death receptor-1 (PD-1) or programmed death-ligand 1 (PD-L1) inhibitor, and a platinum-containing therapy. In 2021, this indication was granted regular approval and Padcev® was granted accelerated approval for patients which are ineligible for cisplatin-containing chemotherapy and have previously received one or more prior lines of therapy. Padcev® is comprised of a fully humanized anti-Nectin4 IgG1κ mAb (AGS-22C3) produced by mammalian (Chinese hamster ovary) cells, and has an approximate DAR of 3.8.
6 .Enhertu® (fam-trastuzumab deruxtecan-nxki), developed by Daichi Sankyo/AstraZeneca, was granted accelerated FDA approval in December 2019 for treatment of adult patients with unresectable or metastatic HER2+ breast cancer who have received two or more prior anti-HER2 based regimens. The ADC is comprised of an anti-HER2 antibody, a protease cleavable tetrapeptide-based linker, and DXd as the drug payload.
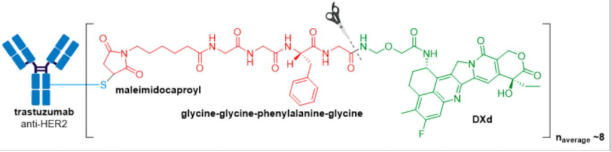
In April 2020, Trodelvy® received accelerated FDA approval for treatment of patients with locally advanced or metastatic triple-negative breast cancer (mTNBC) who have received at least two prior therapies for metastatic disease. Trodelvy® consists of a fully humanized hRS7 IgG1κ antibody targeted against TROP2 (trophoblast antigen 2) conjugated to SN-38, the active metabolite of irinotecan via an acid-sensitive hydrolysable linker called CL2A.
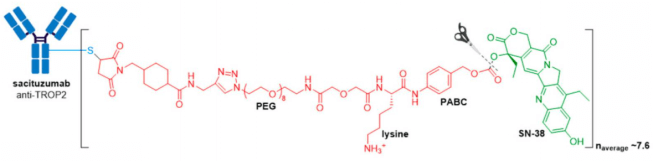
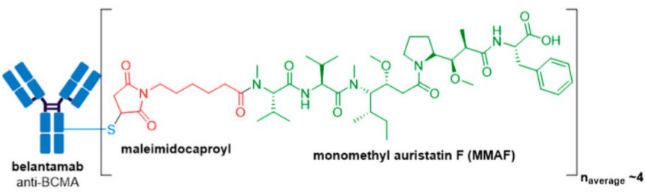
GlaxoSmithKline’s ADC, Blenrep® (belantamab mafodotin-blmf), is the first approved anti-BCMA (B-cell maturation antigen) therapy. It was granted accelerated FDA approval in August 2020 for treatment of adult patients with relapsed or refractory multiple myeloma who have received at least four prior therapies, including an anti-CD38 mAb, a proteasome inhibitor, and an immunomodulatory agent. Blenrep® consists of an afucosylated humanized IgG1 mAb conjugated to the tubulin inhibitor, monomethyl auristatin F (MMAF) via a non-cleavable maleimidocaproyl linker. In addition to MMAF-induced apoptosis, secondary antitumor activity results from tumor cell lysis through ADCC and ADCP effector functions.
Zynlonta® (loncastuximab tesirine-lpyl) developed by ADC Therapeutics is a CD19-directed ADC indicated for treatment of adult patients with relapsed or refractory large B-cell lymphoma after two or more lines of systemic therapy, including diffuse large B-cell lymphoma (DLBCL), not otherwise specified DLBCL arising from low grade lymphoma, and high-grade B-cell lymphoma. It was granted accelerated approval for medical use by the FDA in April 2021. Zynlonta® is composed of a humanized IgG1κ mAb conjugated to SG3199, a cytotoxic pyrrolobenzodiazepine (PBD) dimer alkylating agent, through a protease-cleavable valine-alanine linker.
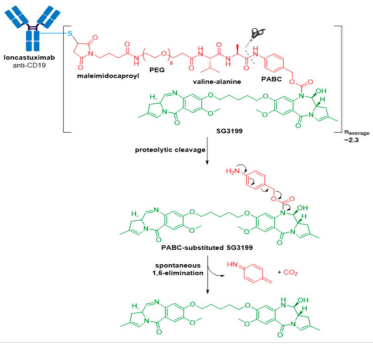
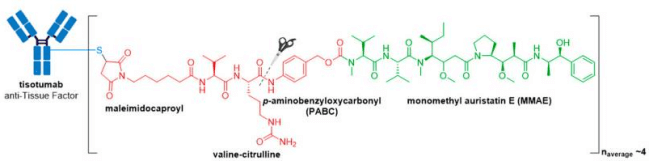
In late September 2021, the FDA granted accelerated approval to Tivdak® (tisotumab vedotin-tftv), deeming it the most recently approved ADC on the market. Tivdak®, co-developed by Seagen and Genmab, is the first and only approved ADC indicated for treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy . Tivdak® is a Tissue Factor (TF) directed ADC comprised of a human anti-TF IgG1κ antibody conjugated to MMAE via the same protease-cleavable mc-vc-PABC linker construct employed in Adcetris®, Polivy®, and Padcev®. As for these previously discussed ADCs, Tivdak® carries an average of four MMAE molecules per mAb. Furthermore, in vitro studies have demonstrated that this ADC also mediates ADCP and ADCC effector functions, thus providing multimodal antitumor activity.
As ADCs have undergone clinical development, it has become clear that the rules applying to standard chemotherapy or antibody-based therapies on their own do not necessarily apply to ADCs. ADCs are modular in nature, with interchangeable components that can be altered in a strategic fashion to improve both their efficacy and toxicity profiles. 1
Reference:
1. Mol Cancer Ther (2021) 20 (5): 885–895.
Product List
Cat No. | Products Name (INN Index) | INN Name | Previous Name | Target | Format | Order |
|---|---|---|---|---|---|---|
Pre-Made Anetumab biosimilar, Whole mAb ADC, Anti-MSLN Antibody: Anti-MPF/SMRP therapeutic antibody |
Anetumab |
NA |
MSLN |
Whole mAb ADC |
||
Pre-Made Aprutumab biosimilar, Whole mAb ADC, Anti-FGFR2 Antibody: Anti-BBDS/BEK/BFR-1/CD332/CEK3/CFD1/ECT1/JWS/K-SAM/KGFR/TK14/TK25 therapeutic antibody |
Aprutumab |
NA |
FGFR2 |
Whole mAb ADC |
||
Pre-Made Azintuxizumab biosimilar, Whole mAb ADC, Anti-SLAMF7/CS1 Antibody: Anti-19A/CD319/CRACC therapeutic antibody |
Azintuxizumab |
NA |
SLAMF7/CS1 |
Whole mAb ADC |
||
Pre-Made Belantamab biosimilar, Whole mAb ADC, Anti-TNFRSF17 Antibody: Anti-BCM/BCMA/CD269/TNFRSF13A therapeutic antibody |
Belantamab |
NA |
TNFRSF17 |
Whole mAb ADC |
||
Pre-Made Brentuximab biosimilar, Whole mAb ADC, Anti-TNFRSF8 Antibody: Anti-CD30/Ki-1/D1S166E therapeutic antibody |
Brentuximab |
NA |
TNFRSF8 |
Whole mAb ADC |
||
Pre-Made Camidanlumab biosimilar, Whole mAb ADC, Anti-IL2RA Antibody: Anti-CD25/IDDM10/IL2R/IMD41/TCGFR/p55 therapeutic antibody |
Camidanlumab |
NA |
IL2RA |
Whole mAb ADC |
||
Pre-Made Cantuzumab biosimilar, Whole mAb ADC, Anti-MUC1 Antibody: Anti-ADMCKD/ADMCKD1/ADTKD2/CA 15-3/CD227/Ca15-3/EMA/H23AG/KL-6/MAM6/MCD/MCKD/MCKD1/SEC/X/ZD/PEM/PEMT/PUM therapeutic antibody |
Cantuzumab |
NA |
MUC1 |
Whole mAb ADC |
||
Pre-Made Cofetuzumab biosimilar, Whole mAb ADC, Anti-PTK7 Antibody: Anti-CCK-4/CCK4 therapeutic antibody |
Cofetuzumab |
NA |
PTK7 |
Whole mAb ADC |
||
Pre-Made Coltuximab biosimilar, Whole mAb ADC, Anti-CD19 Antibody: Anti-B4/CVID3 therapeutic antibody |
Coltuximab |
NA |
CD19 |
Whole mAb ADC |
||
Pre-Made Denintuzumab biosimilar, Whole mAb ADC, Anti-CD19 Antibody: Anti-B4/CVID3 therapeutic antibody |
Denintuzumab |
NA |
CD19 |
Whole mAb ADC |
||
Pre-Made Depatuxizumab biosimilar, Whole mAb ADC, Anti-EGFR Antibody: Anti-ERBB/ERRP/HER1/mENA/ERBB1/PIG61/NISBD2 therapeutic antibody |
Depatuxizumab |
NA |
EGFR |
Whole mAb ADC |
||
Pre-Made Disitamab biosimilar, Whole mAb ADC, Anti-ERBB2/HER2 Antibody: Anti-CD340/neu/MLN 19/NEU/NGL/TKR1/VSCN2 therapeutic antibody |
Disitamab |
NA |
ERBB2 |
Whole mAb ADC |
||
Pre-Made Enapotamab biosimilar, Whole mAb ADC, Anti-AXL Antibody: Anti-ARK/UFO/JTK11/Tyro7 therapeutic antibody |
Enapotamab |
NA |
AXL |
Whole mAb ADC |
||
Pre-Made Enfortumab biosimilar, Whole mAb ADC, Anti-PVRL4/NECTIN4 Antibody: Anti-EDSS1/LNIR/PRR4/nectin-4 therapeutic antibody |
Enfortumab |
NA |
PVRL4 |
Whole mAb ADC |
||
Pre-Made Gemtuzumab biosimilar, Whole mAb ADC, Anti-CD33 Antibody: Anti-p67/SIGLEC3/SIGLEC-3 therapeutic antibody |
Gemtuzumab |
NA |
CD33 |
Whole mAb ADC |
||
Pre-Made Glembatumumab biosimilar, Whole mAb ADC, Anti-GPNMB Antibody: Anti-HGFIN/NMB/PLCA3 therapeutic antibody |
Glembatumumab |
NA |
GPNMB |
Whole mAb ADC |
||
Pre-Made Iladatuzumab biosimilar, Whole mAb ADC, Anti-CD79B Antibody: Anti-AGM6/B29/IGB therapeutic antibody |
Iladatuzumab |
NA |
CD79B |
Whole mAb ADC |
||
Pre-Made Indatuximab biosimilar, Whole mAb ADC, Anti-SDC1 Antibody: Anti-SDC/CD138/SYND1/syndecan therapeutic antibody |
Indatuximab |
NA |
SDC1 |
Whole mAb ADC |
||
Pre-Made Indusatumab biosimilar, Whole mAb ADC, Anti-GUCY2C Antibody: Anti-DIAR6/GC-C/GUC2C/MECILIL/STAR therapeutic antibody |
Indusatumab |
NA |
GUCY2C |
Whole mAb ADC |
||
Pre-Made Inotuzumab biosimilar, Whole mAb ADC, Anti-CD22 Antibody: Anti-SIGLEC2/SIGLEC-2 therapeutic antibody |
Inotuzumab |
NA |
CD22 |
Whole mAb ADC |
||
Pre-Made Labetuzumab biosimilar, Whole mAb ADC, Anti-CEACAM5/CD66e Antibody: Anti-CEA therapeutic antibody |
Labetuzumab |
NA |
CEACAM5 |
Whole mAb ADC |
||
Pre-Made Ladiratuzumab biosimilar, Whole mAb ADC, Anti-SLC39A6 Antibody: Anti-LIV-1/LIV1/ZIP6 therapeutic antibody |
Ladiratuzumab |
NA |
SLC39A6 |
Whole mAb ADC |
||
Pre-Made Laprituximab biosimilar, Whole mAb ADC, Anti-EGFR Antibody: Anti-ERBB/ERRP/HER1/mENA/ERBB1/PIG61/NISBD2 therapeutic antibody |
Laprituximab |
NA |
EGFR |
Whole mAb ADC |
||
Pre-Made Lifastuzumab biosimilar, Whole mAb ADC, Anti-SLC34A2 Antibody: Anti-NAPI-3B/NAPI-IIb/NPTIIb/PULAM therapeutic antibody |
Lifastuzumab |
NA |
SLC34A2 |
Whole mAb ADC |
||
Pre-Made Lilotomab biosimilar, Whole mAb ADC, Anti-CD37 Antibody: Anti-GP52-40/TSPAN26 therapeutic antibody |
Lilotomab |
NA |
CD37 |
Whole mAb ADC |
||
Pre-Made Loncastuximab biosimilar, Whole mAb ADC, Anti-CD19 Antibody: Anti-B4/CVID3 therapeutic antibody |
Loncastuximab |
NA |
CD19 |
Whole mAb ADC |
||
Pre-Made Lorvotuzumab biosimilar, Whole mAb ADC, Anti-NCAM1 Antibody: Anti-CD56/NCAM/MSK39 therapeutic antibody |
Lorvotuzumab |
NA |
NCAM1 |
Whole mAb ADC |
||
Pre-Made Losatuxizumab biosimilar, Whole mAb ADC, Anti-EGFR Antibody: Anti-ERBB/ERRP/HER1/mENA/ERBB1/PIG61/NISBD2 therapeutic antibody |
Losatuxizumab |
NA |
EGFR |
Whole mAb ADC |
||
Pre-Made Lupartumab biosimilar, Whole mAb ADC, Anti-LYPD3 Antibody: Anti-C4.4A therapeutic antibody |
Lupartumab |
NA |
LYPD3 |
Whole mAb ADC |
||
Pre-Made Milatuzumab biosimilar, Whole mAb, Anti-CD74 Antibody: Anti-CLIP/DHLAG/HLADG/II/Ia-GAMMA/p33 therapeutic antibody |
Milatuzumab |
NA |
CD74 |
Whole mAb ADC |
||
Pre-Made Mirvetuximab biosimilar, Whole mAb ADC, Anti-FOLR1 Antibody: Anti-FBP/FOLR/FRalpha/NCFTD therapeutic antibody |
Mirvetuximab |
NA |
FOLR1 |
Whole mAb ADC |
||
Pre-Made Naratuximab biosimilar, Whole mAb ADC, Anti-CD37 Antibody: Anti-GP52-40/TSPAN26 therapeutic antibody |
Naratuximab |
NA |
CD37 |
Whole mAb ADC |
||
Pre-Made Pelgifatamab biosimilar, Whole mAb ADC, Anti-FOLH1/GCPII Antibody: Anti-FGCP/FOLH/GCP2/NAALAD1/PSM/PSMA/mGCP therapeutic antibody |
Pelgifatamab |
NA |
FOLH1 |
Whole mAb ADC |
||
Pre-Made Pinatuzumab biosimilar, Whole mAb ADC, Anti-CD22 Antibody: Anti-SIGLEC2/SIGLEC-2 therapeutic antibody |
Pinatuzumab |
NA |
CD22 |
Whole mAb ADC |
||
Pre-Made Polatuzumab biosimilar, Whole mAb ADC, Anti-CD79B Antibody: Anti-AGM6/B29/IGB therapeutic antibody |
Polatuzumab |
NA |
CD79B |
Whole mAb ADC |
||
Pre-Made Rolinsatamab biosimilar, Whole mAb ADC, Anti-PRLR Antibody: Anti-HPRL/MFAB/RI-PRLR/hPRLrI therapeutic antibody |
Rolinsatamab |
NA |
PRLR |
Whole mAb ADC |
||
Pre-Made Rovalpituzumab biosimilar, Whole mAb ADC, Anti-DLL3 Antibody: Anti-SCDO1 therapeutic antibody |
Rovalpituzumab |
NA |
DLL3 |
Whole mAb ADC |
||
Pre-Made Sacituzumab biosimilar, Whole mAb ADC, Anti-TACSTD2 Antibody: Anti-EGP-1/EGP1/GA733-1/GA7331/GP50/M1S1/TROP2 therapeutic antibody |
Sacituzumab |
NA |
TACSTD2 |
Whole mAb ADC |
||
Pre-Made Samrotamab biosimilar, Whole mAb ADC, Anti-LRRC15 Antibody: Anti-LIB therapeutic antibody |
Samrotamab |
NA |
LRRC15 |
Whole mAb ADC |
||
Pre-Made Serclutamab biosimilar, Whole mAb ADC, Anti-EGFR Antibody: Anti-ERBB/ERRP/HER1/mENA/ERBB1/PIG61/NISBD2 therapeutic antibody |
Serclutamab |
NA |
EGFR |
Whole mAb ADC |
||
Pre-Made Sirtratumab biosimilar, Whole mAb ADC, Anti-SLITRK6 Antibody: Anti-DFNMYP therapeutic antibody |
Sirtratumab |
NA |
SLITRK6 |
Whole mAb ADC |
||
Pre-Made Sofituzumab biosimilar, Whole mAb ADC, Anti-MUC16 Antibody: Anti-CA125 therapeutic antibody |
Sofituzumab |
NA |
MUC16 |
Whole mAb ADC |
||
Pre-Made Tabituximab biosimilar, Whole mAb ADC, Anti-FZD10 Antibody: Anti-CD350/FZ-10/Fz10/FzE7/hFz10 therapeutic antibody |
Tabituximab |
NA |
FZD10 |
Whole mAb ADC |
||
Pre-Made Tamrintamab biosimilar, Whole mAb ADC, Anti-DPEP3 Antibody: Anti-MBD3 therapeutic antibody |
Tamrintamab |
NA |
DPEP3 |
Whole mAb ADC |
||
Pre-Made Telisotuzumab biosimilar, Whole mAb ADC, Anti-MET Antibody: Anti-AUTS9/DFNB97/HGFR/RCCP2/c-Met therapeutic antibody |
Telisotuzumab |
NA |
MET |
Whole mAb ADC |
||
Pre-Made Vadastuximab biosimilar, Whole mAb ADC, Anti-CD33 Antibody: Anti-p67/SIGLEC3/SIGLEC-3 therapeutic antibody |
Vadastuximab |
NA |
CD33 |
Whole mAb ADC |
||
Pre-Made Vandortuzumab biosimilar, Whole mAb ADC, Anti-STEAP1 Antibody: Anti-PRSS24/STEAP therapeutic antibody |
Vandortuzumab |
NA |
STEAP1 |
Whole mAb ADC |
||
Pre-Made Vorsetuzumab biosimilar, Whole mAb ADC, Anti-CD70/CD27-L Antibody: Anti-CD27L/LPFS3/CD27LG/TNFSF7/TNLG8A therapeutic antibody |
Vorsetuzumab |
NA |
CD70 |
Whole mAb ADC |
||
Pre-Made Tabituximab Barzuxetan Biosimilar, Whole Mab Adc, Anti-Fzd10 Antibody: Anti-CD350/FZ-10/Fz10/FzE7/hFz10 therapeutic antibody Drug Conjugate |
tabituximab barzuxetan |
NA |
FZD10 |
Whole mAb ADC |
||
Pre-Made Tamrintamab Pamozirine Biosimilar, Whole Mab Adc, Anti-Dpep3 Antibody: Anti-MBD3 therapeutic antibody Drug Conjugate |
tamrintamab pamozirine |
NA |
DPEP3 |
Whole mAb ADC |
Reference
1. Mol Cancer Ther (2021) 20 (5): 885–895.
View the Knowledge base of Antibody-drug Conjugate (ADC):
– What is antibody-drug conjugate (ADC)?
– Antibody-drug conjugate (ADC) in clinical application (Approved/BLA, phaseI/II/III)
– Main elements of antibody-drug conjugate (ADC): Antibodies and their targets
– Main elements of antibody-drug conjugate (ADC):Linker (cleavable/non-cleavable, structure and mechanism)
– Main elements of antibody-drug conjugate (ADC):Toxins/Payloads (Classification and function)
– Toxins/Payloads (Classification and function) of Microtubule destroying drug
– Toxins/Payloads (Classification and function) of DNA damage drugs
– Toxins/Payloads (Classification and function) of Innovative drugs
– Biological coupling technology Chemical based specific in situ antibody modification
– Endogenous coupling of amino acids and Disulfide re bridging strategy
– Glycan coupling
– Site specific biological coupling of engineered antibodies and Enzymatic method
– Biological coupling with engineered unnatural amino acids
– Review for ADC production, quality control and functional assay
– Product data of ADC




